-
CENTRES
Progammes & Centres
Location
 PDF Download
PDF Download 
Erfan Ibrahim and Shahid Jameel, “Understanding Disease and Shaping Public Health Policy in a Time of Pandemic,” ORF Issue Brief No. 358, May 2020, Observer Research Foundation.
Introduction
A new virus emerged in China in late 2019, spread across the globe within a few months, and was declared a pandemic by the World Health Organization (WHO) in the middle of March 2020.[1] Due to its similarity to the severe acute respiratory syndrome coronavirus (SARS-CoV) that emerged in 2002-03, the new virus was called SARS-CoV-2, and the disease, ‘coronavirus infectious disease 2019’ or COVID-19. As of 2 May 2020, there are over 3.4 million confirmed cases and about 244,000 deaths reported from 187 countries. Of these, the United States (US) has recorded over 1.1 million cases, and seven others–Spain, Italy, the United Kingdom (UK), France, Germany, Turkey and Russia—each recording over 100,000 cases; together these eight countries contribute almost 70 percent of all cases.[2]India has recorded almost 40,000 confirmed cases and 1,323 deaths.[3]
There is currently no specific cure for COVID-19, and the forecast for a vaccine puts it at 12 to 18 months, at least, at the time of writing. Although population infection rates are not yet available, these are likely to be well below the 60 percent threshold required for developing “herd immunity” to COVID-19.[4] With no opportunity for prophylactic or therapeutic mitigation, and with high mortality rates in the elderly and those with co-morbidities, the only way of preventing the spread of the highly infectious disease is to avoid people already infected. This becomes difficult when a large fraction of infected people shows mild symptoms, if at all.
National and state governments across the globe have enforced various levels of “shelter in place” policies to reduce the rate of community transmission. In India, this first took the form of a 21-day nationwide lockdown beginning on 25 March, which was later extended for another 19 days. Another two-week extension has been announced with regional flexibility based on the local infection situation. These measures are designed to reduce the number of critically ill COVID-19 patients entering hospitals and overwhelming facilities, and to give authorities time to strengthen infrastructure (e.g. Personal Protective Equipment, ventilators, ICU beds, acute care hospital beds). However, these measures have also resulted in major economic losses for nations and individuals, less healthcare support for other diseases, and widespread hunger and anxiety amongst the vulnerable sections of society.
After three to five weeks of lockdown enforcement, several nations, including India, have started experiencing a slowdown in the spread of COVID-19.[5] However, new cases continue to be recorded and the cumulative death figures keep rising in almost every country across the globe.
This brief provides an overview of COVID-19, in the context of making recommendations to assist public health officials and policymakers in finding measures for arresting the pandemic.
The Science of COVID-19
SARS-CoV-2 belongs to the larger Coronavirus family of animal viruses, which are so named due to their crown-like (“corona”) appearance when magnified by an electron microscope. Six other coronaviruses are known to cause disease in humans: SARS-CoV, Middle East Respiratory Syndrome coronavirus (MERS-CoV), 229E, OC43, 229E, and NL63. Of these, the last four are responsible for about 20-30 percent of common colds. Besides humans, other mammals including bats harbour a large number of coronaviruses, and previous studies have shown some of these bat coronaviruses to have the potential to infect humans. SARS-CoV-2 appears to have evolved from one of those.[6]
The SARS-CoV-2 virus particle is about 100 nm (0.1 micrometer or micron) in size, about a million times smaller than the cells it infects.[7] Its genome is a ribonucleic acid (RNA) of about 30,000 nucleotides (about 100,000 times smaller than the human genome) that is tightly packed over a scaffold of the viral nucleocapsid protein.[8] This makes the core of the virus particle, which is then packaged inside a membrane made of lipids and three other proteins – the Spike protein that sticks out to give a crown-like appearance, and the Envelope and Membrane proteins that sit between the spikes (See Figure 1). The virus membrane breaks up with soap and water, which is why handwashing protects from infection.
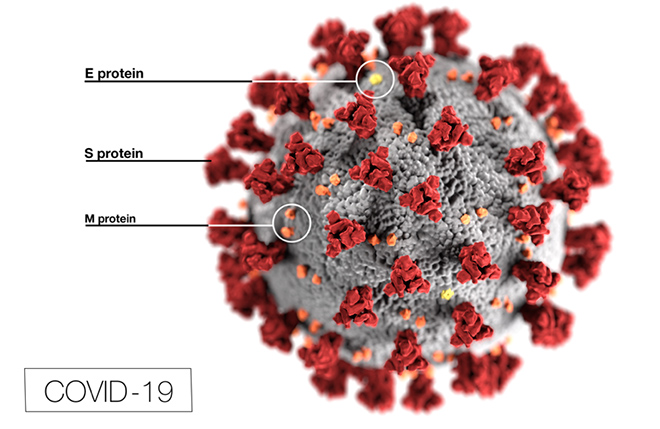
The virus enters the body most commonly through the mouth, nose or eyes. It is typically carried in the saliva or nasal secretion of the infected person.[10] The transfer can occur either if the infected person coughs or sneezes within three feet of an unprotected person, or if the unprotected person touches the infected person or a surface they have recently touched or sneezed on, and then touches their face, mouth or eyes. The virus can remain infectious on surfaces for hours to days, and that is why regularly cleaning these with disinfectants is a simple and effective mitigation strategy.[11]
Understanding the virus life cycle helps in understanding why health and medical experts are using or testing certain drugs to treat COVID-19 patients.[12] The first contact between the virus and target cells is made when the Spike protein of the virus binds to a protein called ACE2 on the surface of human cells, particularly those in the throat and nasopharynx. This step can be inhibited by antibodies to the Spike protein found in the plasma of recovered patients and is the basis for plasma therapy.[13] Once attached, a protease (an enzyme that cuts other proteins) on the cell surface cuts the Spike protein to trigger the fusion of virus and cell membranes and delivery of the particle inside the cell. An inhibitor of this protease is yet another target for developing antivirals.[14] The particle delivery takes place through subcellular compartments called endosomes, which is compromised by chloroquine and hydroxychloroquine. For this reason, these anti-malaria drugs also show variable efficacy against many viruses.[15]
The viral RNA is released and is converted by the cell’s machinery into viral proteins, some of which are processed by a viral protease, which can be another target. Some anti-HIV protease inhibitor drugs have shown limited efficacy in COVID-19 patients.[16] The viral RNA is multiplied by another enzyme called RNA dependent RNA polymerase (RdRp) or replicase, which is yet another drug target. A drug called Remdesivir initially developed for Ebola virus and another called Favipiravir developed for influenza virus, are now being tested against COVID-19.[17] The replicase produces more RNA copies and through it more proteins. These rounds of amplification produce many new virus particles that are released from infected cells.[18]
Based on in vitro studies, the residence time of the virus on cells is approximately 10 minutes, followed by an eclipse period of around 10 hours during which the cell is replicating the virus. The burst size, i.e. number of new virus particles produced by each infected cell, is estimated to be ~ 103.[19] If the immune system does not control SARS-CoV-2 in the throat and nasopharynx, it marches down the windpipe to attack the lungs, where it mainly infects pneumocytes that are responsible for oxygen exchange with blood but are also rich in ACE2 receptors.[20] The immune system summons its defences and the ensuing fight leaves behind a mix of fluid and dead cells. This causes oxygen deprivation and is the underlying pathology of pneumonia characterised by cough, fever and shallow breathing. Some patients go into a severe form of pneumonia, called acute respiratory distress syndrome (ARDS). This is the critical phase when patients require oxygen support and ventilation.[21] Besides this lung pathology, COVID-19 patients also show other curious features to various degrees that are not fully understood at this time. These include blood clots, as well as heart, liver and kidney damage, and even neurological symptoms. The virus possibly multiplies in the gut as well, with associated diarrhoea and is shed in stool for extended periods of time.[22]
At the time of writing, the test widely used is called a reverse transcription polymerase chain reaction (RT-PCR) test, which detects the viral genetic material (RNA). Nasal aspirates and throat swabs are commonly used since these can be non-invasively sampled, but lung aspirates, sputum, blood and stool have also been used to detect the virus. The other type of test looks for antibodies against the virus made by infected persons. Two types of antibodies – IgM and IgG—are detected in a drop of blood from a finger prick.
The differences between these two tests and their significance are important to understand testing strategies that are useful for making decisions during and after the pandemic. A patient shows positive by the PCR test on the day symptoms appear, and possibly two to three days prior.[23] The windows for IgM and IgG antibodies are about seven to 40 days and 10 to >50 days, respectively, after the appearance of symptoms.[24] This is illustrated in Figure 2. Therefore, antibody tests are not confirmatory as they would miss early infections. A positive antibody test may indicate either an ongoing infection (IgM+IgG- or IgM+IgG+) or a past infection (IgM-IgG+). More work is needed to understand if IgG antibodies will protect from future infections, what might be their required levels and for how long. The tests available at this time are only qualitative and not quantitative – they indicate the absence or presence of antibodies, not their concentration in blood.
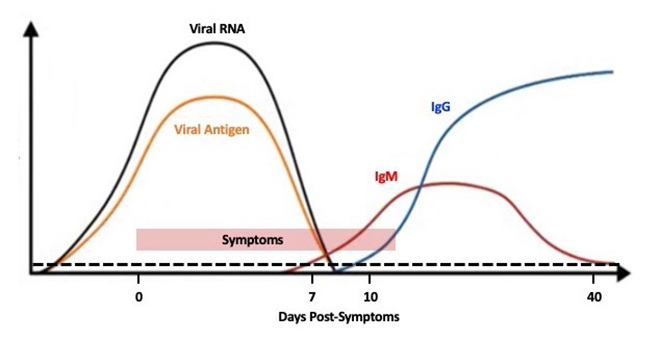
While the PCR test takes a couple of hours and uses special laboratories and equipment, the antibody test takes just minutes and can be used for point-of-care (POC) testing. This also reflects in their costs – PCR test for about $50 (Rs. 4500); antibody test for under $10 (Rs 500-1000). The utility of a PCR test is in confirming individual diagnosis and that of the antibody test in assessing population exposure.
The Foundation for Innovative New Diagnostics, a global non-profit organisation, is evaluating five new antigen detection rapid diagnostic tests (RDTs), 27 new antibody detection RDTs, and seven new antibody measurement ELISA tests.[26]The last one determines antibody levels, which would be highly useful in post-lockdown and post-pandemic assessments.
There are currently no approved drugs to treat COVID-19, but various experimental therapies have been tried with limited success and are the subject of larger trials. These include the HIV protease inhibitors and drugs developed against Ebola and flu viruses, described above.[27] The US Food and Drug Administration (FDA) and Indian regulators have also allowed hydroxychloroquine to be tried on patients, and in the latter case, to be used as preventive medication for frontline healthcare workers and COVID-19 contacts.[28]
Not all drugs work by targeting the virus. Some like interferons work by enhancing the innate immune response and have shown limited efficacy in COVID-19 patients. The virus can also cause damage to lungs by hyperactivating the immune system’s inflammatory response. A molecule called interleukin-6 is involved and an antibody (Actemra; Tocilizumab) that blocks its action has shown therapeutic effect.[29] Scientists have also identified many existing drugs and novel chemical entities, some already used to treat other diseases. Repurposing them to treat COVID-19, would be faster than developing a completely new drug.[30]
Around 115 different candidate vaccines against COVID-19 are in development, of which six are in Phase 1 human clinical trials. These include – mRNA vaccine, Moderna, USA; adenovirus 5 vectored Spike protein, CanSino Biologicals, China; DNA encoding the Spike protein, Inovio Pharmaceuticals, USA; chimpanzee adenovirus vectored Spike protein, Oxford University, UK; and two modified cell vaccines, Shenzhen GIMI, China.[31]
It takes years to develop a vaccine, first in the laboratory, then manufacturing stable and highly pure products to be tested in animals and humans. The COVID-19 vaccine has been fast-tracked into humans with parallel testing in animals. Still, it may take about 12-18 months for the clinical trials to show safety and efficacy. Perhaps the most ambitious is the Oxford vaccine trial, which plans to enroll over 1100 people directly into an efficacy study, and projects results within six months.[32]
Three Indian companies are developing COVID-19 vaccines with overseas partnerships. These include the Serum Institute, Pune and Codagenix, USA for a genetically attenuated vaccine; Bharat Biotech, Hyderabad and University of Wisconsin, USA for a nasal vaccine based on the flu-mist platform; and Premas Biotech, Manesar and Akers Biosciences, USA for recombinant viral proteins.[33]
It remains to be seen which of these vaccines will eventually be found to work. However, the speed at which multiple vaccines have moved into human trials within two to three months of virus identification is remarkable and unprecedented.
The Management of a Pandemic
The following paragraphs make a comparison of the different COVID-19 forecasting models to understand future trends and offer a comprehensive plan that is informed by science and incorporates compassion and socioeconomic realities.
Infection rates and the cumulative death count from COVID-19 are increasing daily. A majority of deaths are in older patients (60+ years) with co-morbidities, but younger people are also getting infected in large numbers, albeit with reduced mortality. The daily confirmed COVID-19 cases and death curves are not flattening at the rate commensurate with a two- to three-week lifecycle of the virus in places with long-duration “shelter in place” policies such as in New York State and California in the United States. At the same time, hospitalisation levels of COVID-19 patients are dropping in these states since mid-April. This suggests that the “shelter in place” strategy may be reaching the point of diminishing returns in these areas because either the new confirmed cases are the result of antibody tests on people that are no longer infectious, or new people without serious symptoms are getting infected because they are not able to maintain effective social distancing in their living conditions.[34] [35] These locations are unlikely to benefit any further from extending the interventions for much longer. Instead, there would be severe economic and social consequences for a restless population that is without work, money and food, and no credible recovery path.
Other US states such as Florida, Georgia, Maryland, North Carolina and Pennsylvania that invoked the “shelter in place” strategy later in March or early April 2020 are witnessing growing numbers of confirmed COVID-19 cases, death rates and hospitalisation levels.[36] India initially invoked a nationwide lockdown on 25 March 2020 for 21 days, which was first extended for 19 more days till 3 May 2020, and subsequently another 14 days till 17 May 2020.
1. COVID-19 mortality rates are of the same order of magnitude as seasonal flu, and not the 2-10 percent being shown by media.
In late March 2020, the authors of this brief estimated the COVID-19 population mortality rate to be 0.5 percent. This was calculated by taking the average clinical mortality of 2.5 percent from US hospitals at the time, and dividing it by five, because for every person that approached a hospital with life-threatening COVID-19 symptoms, four others were sent home without testing due to mild symptoms.[37] Additionally, the average time from onset of illness to death was estimated to be about 18 days based on a study of 54 non-surviving patients in Wuhan China (actual range was 15-22 days).[38] The “Hybrid Model” therefore takes the cumulative death count on a given day and multiplies it by a factor of 200 to predict the cumulative COVID-19 infection count 18 days earlier. Knowing the doubling time of the outbreak in a given area, the Hybrid Model allows approximation of the current cumulative numbers of infections.
In early April 2020, a Lancet report estimated the COVID-19 mortality rate to be 0.66 percent based on the fact that several patients never enter the healthcare system and are not tested because they are either asymptomatic, do not have life-threatening symptoms, are under or uninsured, and recover in isolation at home.[39] Another study by the Stanford School of Medicine, USA in early April 2020 used random antibody testing of 3300 residents in Santa Clara County (California, USA) to suggest a 0.12 to 0.2 percent mortality rate for COVID-19.[40] This was validated by antibody testing in Los Angeles (California, USA) in the 3rd week of April.[41] New York State released results from an antibody test on 22 April 2020 that suggested the mortality rate from COVID-19 to be in the 0.5 percent to 0.78 percent range, in agreement with the Lancet Model and the Hybrid Model.[42]
2. The numbers of deaths attributed to COVID-19 are not accurate in some countries. For instance, in the United States and Belgium, hospitals are lumping together confirmed COVID-19 deaths with “probable cases” which could be a result of other respiratory illnesses.[43] [44] There is neither an autopsy report nor a valid COVID-19 virus test to confirm these “probable cases”. The US Centers for Disease Control has allowed hospitals in the country to include “probable cases” for COVID-19 in the official death count as of April 14, 2020. The cause of death that is entered on a death certificate in a hospital is typically based on the judgment of the attending physician. There is considerable room for human error in this decision process in the absence of a valid COVID-19 virus test result or a formal autopsy. Additionally, many epidemiological symptoms of COVID-19 resemble those of other respiratory illnesses and can be easily confused when the patient volumes are high and the time to make a decision is short. This will artificially increase the death count and overestimate infection in the community. It also delays flattening of the death curve for COVID-19 because patients who may have died from flu, asthma, or other respiratory ailments will keep inflating the daily death numbers well past the time when COVID-19 infection levels are low enough to resume contact tracing of cases in a community. At the same time, COVID-19 deaths at home might not be accounted for completely because there is no formal process for reporting these deaths in the official tally. All this adds to the uncertainty in the official death counts from COVID-19 and makes it more challenging to estimate the mortality rate from the disease.
3. The “new cases” for COVID-19 being entered into global records say nothing about when the patient was infected. PCR tests measure the presence of viral RNA and not the infectious nature of the virus. These are positive as late as 30-35 days from the onset of symptoms, when antibodies have developed, and the virus is no longer expected to be viable nor is the patient infectious.[45] This lack of a biological timeline with new positive COVID-19 tests will also create an artificial delay in flattening of the infection curve well beyond the biological timeline for the spread of the infection in a community.
All of the above problems will create pressure on governments to keep extending the “shelter in place” or lockdown beyond their useful time span.
COVID-19 Cumulative Infection Predictive Models: A Brief Comparison
A comparison of the Lancet, Hybrid and Stanford models for COVID-19 and the clinical data of cumulative infections and deaths is given in Figures 3 and 4 for the US and India, respectively.[46] All these models estimate the number of infections to be much higher in both the US and India compared to confirmed cases. Additionally, all cumulative infection graphs for the US (Figure 3) show a slight flattening of the curve starting around 31 March 2020, more than a week after “shelter in place” policies were enforced across several states with further flattening beyond 21 April 2020. Similarly, for India (Figure 4) a slight flattening of the curve is observed starting around 7 April 2020 i.e. 13 days after a nationwide lockdown was enforced and testing became more readily available.
Figure 3: Cumulative Infection Graph, USA.
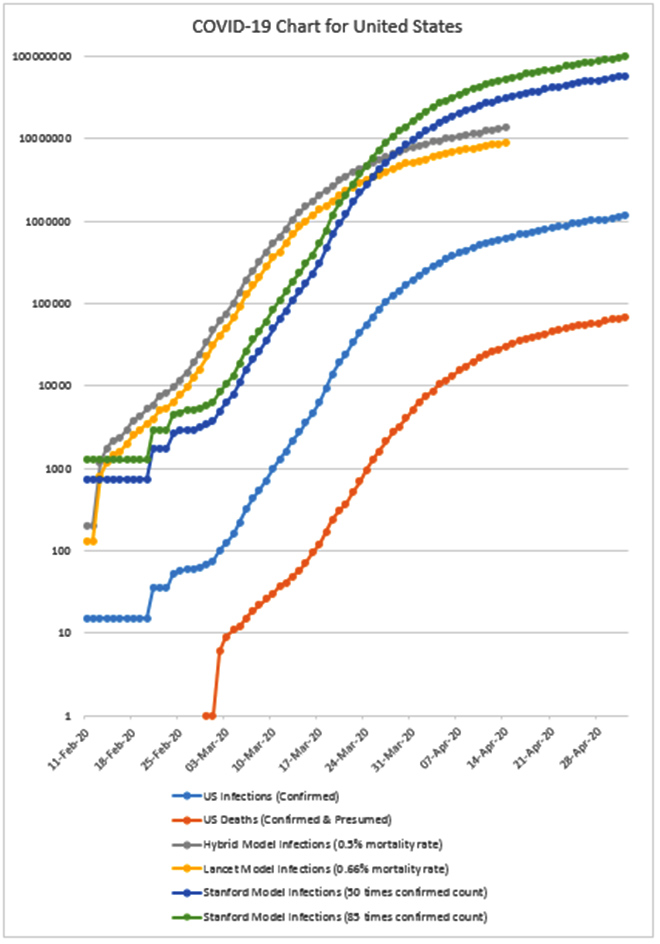
Figure 4: Cumulative Infection Graph, India.
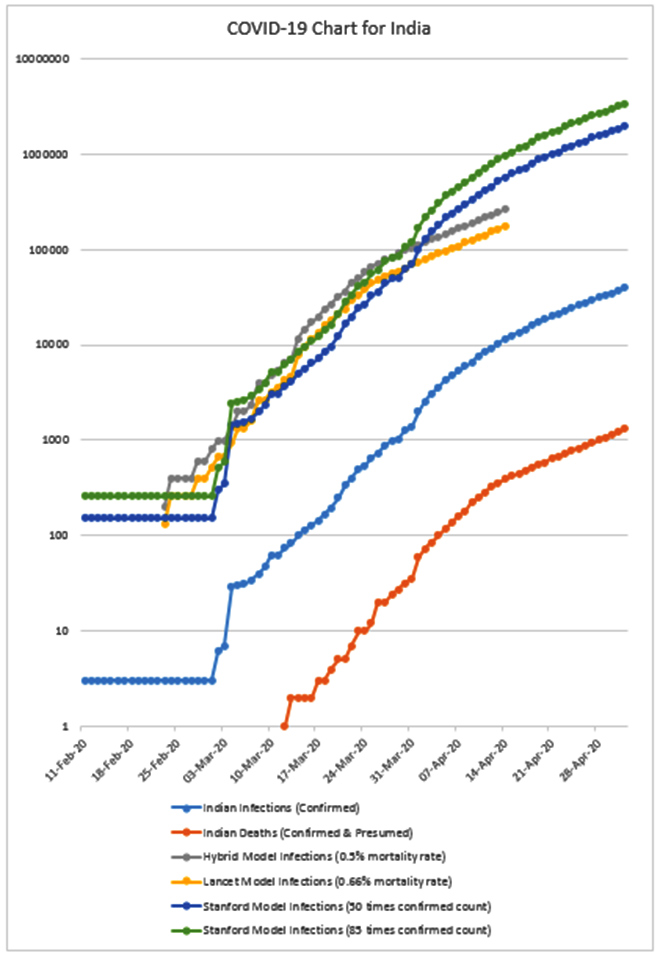
Four key models have been used to forecast the number of COVID-19 infections in India. If these are compared to the Lancet, Hybrid and Stanford models, the following are the conclusions that can be drawn:
As of 2 May 2020, there are 1,323 deaths and almost 40,000 cumulative COVID-19 cases in India, with both figures rising daily despite the continuous lockdown since 25 March 2020. The 2-10 percent mortality rate projected by global media is the case fatality rate based on confirmed positive cases in various countries. This rate is five to 80 times greater than the value that scientific research is determining using random testing of the population for COVID-19, thereby increasing the level of alarm globally and enabling extreme measures to be considered for mitigating the “perceived” life risks.
Balancing Social Science with Physical Science
Social distancing through lockdown is effective in dramatically slowing down community transmission of infectious diseases such as COVID-19. However, it should be considered as a last resort to contain a virus when contact tracing of cases, facial masks and hand hygiene recommendations are not proving to be sufficient. This is because lockdowns have a devastating effect on small businesses, the service industry, daily wage earners, and the poor. If lockdowns are prolonged beyond their ability to contain an infectious disease, they put disproportionate financial, social, psychological and emotional burdens on the most vulnerable segments of the population.
Balancing social science with physical science means monitoring the biological spread of the virus in the community and ending lockdowns as soon as the number of new cases and hospitalisation levels drop to a level where contact tracing of cases can resume and the public can be protected with facial masks and hand hygiene. If adulterated metrics are used to artificially exaggerate the level of active infection in a community, the extension of lockdowns will do more harm than good in maintaining the health and wellbeing of the community.
Politicians, public health officials and business leaders need to use objective scientific evidence to assess the status of the pandemic at any point in time and create policies that do not harm vulnerable segments of the population. Social unrest, starvation, lack of adequate healthcare and economically motivated crime may create mortality rates far higher than the rates from COVID-19 infection several weeks into the lockdown.
Recommendations
This brief offers five key recommendations for public health policy and political leadership to consider in India and globally.
Conclusion
The fast pace at which the COVID-19 pandemic has spread globally, the death and fear it has created is unprecedented. The tragedy of the ongoing pandemic can be measured in terms of human life, economic loss, physical trauma, emotional stress and social disorder. It is critical for political, public health policy and economic leaders to work together to get the global population out of this abyss.
This brief aimed to assess the current situation, offered an overview of the science of the disease, compared various forecasting models for COVID-19 infection levels and mortality rates, and listed five key recommendations for how India and other nations can potentially come out of the lockdown while maintaining social, economic and political stability.
The COVID-19 pandemic will not be the last of its kind, and the world cannot afford to go into a lockdown each time a crisis of such magnitude happens. Lessons learned from COVID-19 should enable the world to better manage future pandemics.
About the authors
Dr. Erfan Ibrahim has worked in advanced nuclear technology, mathematical modeling, networking, telecommunications, smart grid and cybersecurity during his 33-year professional career. He holds a BS Honors in Physics from Syracuse University, an MS in Mechanical Engineering from University of Texas at Austin and a PhD in Nuclear Engineering from University of California, Berkeley.
Dr. Shahid Jameel established and for 25 years led the Virology Group at the International Centre for Genetic Engineering and Biotechnology, New Delhi. He is currently CEO of the DBT/Wellcome Trust India Alliance, a biomedical research public charity based in India.
Endnotes
[a] The SEIR Model puts the population into four groups: susceptible (S), exposed (E), infected (I), and recovered or removed (i.e. dead) (R).
[b] The SIR Model puts the population into three groups: susceptible (S), infected (I), and recovered or removed (i.e. dead) (R).
[1] World Health Organization. Director-General’s Speech, 11 March 2020.
[2] Enshen Dong, Hongru Du and Lauren Gardner. An interactive web-based dashboard to track COVID-19 in real time. Lancet Infect Dis; published online Feb 19, 2020.
[3] Ministry of Health and Family Welfare, Government of India. Downloaded at 12:45 pm on May 2, 2020.
[4] Daniel M. Altman, Daniel C. Douek, and Rosemary J. Boyton. What policy makers need to know about COVID-19 protective immunity. The Lancet April 27, 2020.
[5] Arghya Das, Abhishek Dhar, Srashti Goyal, et al. COVID-19 data analysis based commentary. Accessed May 2, 2020.
[6] Catharine I. Paules, Hillary D. Marston and Anthony S. Fauci. Coronavirus Infections –More than just the common cold. JAMA. 2020; 323(8):707-708. Doi:10.1001/jama.2020.0757 [herein after: Catharine, Hillary and Anthony. Corona Infection]
[7] Ibid
[8] Yinon M. Bar-On, Avi Flamholz, Rob Phillips and Ron Milo. SARS-CoV-2 (COVID-19) by the numbers. eLife. 2020; 9:e57309 DOI: 10.7554/eLife.57309 [Herein after: Yinon, Avi, Rob, Ron. SARS-CoV2]
[9] Centers for Disease Control and Prevention, USA. Image Library. Accessed May 2, 2020.
[10] Catharine, Hillary and Anthony. Corona Infection
[11] Neeltje van Doremalen, Trenton Bushmaker, Dylan H. Morris, et al. Aerosol and surface stability of SARS-CoV-2 as compared to SARS-CoV-1. New Engl J Med.March 17, 2020; DOI: 10.1056/NEJMc2004973. [Herein after: Neeltje, Trenton, Dylan. Aerosol and surface stability of SARS-CoV2 ]
[12] Ibid
[13] Ibid
[14] Ibid
[15] Ibid
[16] Ibid
[17] Ibid
[18] “Understanding SARS-CoV-2 and the drugs that might lessen its power”, The Economist, March 12, 2020.
[19] Yinon, Avi, Rob, Ron. SARS-CoV2
[20] Ibid
[21] Ibid
[22] Meredith Wadman, Jennifer Couzin-Frankel, Jocelyn Kaiser, Catherine Matacic. How does coronavirus kill? Clinicians trace a ferocious rampage through the body, from brain to toes. Science. April 17, 2020;
[23] Ibid
[24] Wenting Tan, Yanqiu Lu, Juan Zhang, et al. Viral kinetics and antibody responses in patients with COVID-19. medRxiv. March 26, 2020.
[25] Centers for Disease Control and Prevention, USA. Image Library. Accessed May 2, 2020.
[26] Foundation for Innovative New Diagnostics. Evaluation Update: SARS-CoV-2 immunoassays. Accessed May 2, 2020.
[27] Tanya Lewis. Here’s what we know about the most touted drugs tested for COVID-19. Scientific American. April 16, 2020;
[28] Sahaj Rathi, Pranav Ish, Ashwini Kalantri and Shriprakash Kalantri. Hydroxychloquine prophylaxis for COVID-19 contacts in India. The Lancet. April 17, 2020.
[29] Binqing Fu, Xiaoling Xu and Haiming Wei. Why tocilizumab could be an effective treatment for severe COVID-19? J Translational Med. 2020; 18:164.
[30] Carl Zimmer, “Scientists Identify 69 Drugs to Test Against the Coronavirus”, The New York Times, March 22, 2020.
[31] Tung Thanh Le, Zackarias Andreadakis, Arun Kumar, et al. The Covid-19 Vaccine Development Landscape. Nature Reviews Drug Discovery. 2020; 19:305-306.
[32] David D. Kirkpartick, “In race for a coronavirus vaccine, an Oxford group leaps ahead”, The New York Times, April 27, 2020.
[33] Namita Kohli, “Team spirit – How many are joining hands to develop a vaccine for COVID-19”, The Week, April 23, 2020.
[34] New York City Health Department. COVID-19: data. Accessed May 2, 2020.
[35] Santa Clara Country Public Health Emergency Operations Center. Coronavirus (COVID-19) data dashboard. Accessed May 2, 2020.
[36] Centers for Disease Control and Prevention, USA. Geographic Differences in COVID-19 Cases, Deaths, and Incidence — United States, February 12–April 7, 2020. MMWR Morb Mrtal Wkly Rep. 2020; 69:465-471. DOI:
[37] David Price, “Protecting families during COVID-19 pandemic”, YouTube video, April 1, 2020.
[38] Fei Zhou, Ting Yu, Ronghui Du, et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. The Lancet, 2020; 395:1054-1062. DOI: 10.1016/S0140-6736(20)30566-3
[39] Robert Verity, Lucy C. Okell, Ilaria Dorigatti, et al. 2020. Estimates of severity of coronavirus disease 2019: a model-based analysis. The Lancet; March 30, 2020.
[40] Eran Bendavid, Bianca Mulaney, Neeraj Sood, et al. COVID-19 antibody seroprevalence in Santa Clara County, California. medRxiv. April 30, 2020.
[41] Jacob Sullum, “L.A. County Antibody Tests Suggest the Fatality Rate for COVID-19 Is Much Lower Than People Feared”, Reason, April 20, 2020.
[42] J. David Goodman and Michael Rothfeld, “1 in 5 New Yorkers may have had COVID-19, antibody tests suggest”, The New York Times, April 23, 2020.
[43] Centers for Disease Control and Prevention, USA. “FAQ: COVID-19 data and surveillance”, April 17, 2020.
[44] Barbara Moens, “Why is Belgium’s death toll so high?”, Politico, April 19, 2020.
[45] Neeltje, Trenton, Dylan. Aerosol and surface stability of SARS-CoV2
[46] Worldometers, Covid-19 Coronavirus Pandemic. Accessed May 2, 2020.
[47] Sandip Mandal, Tarun Bhatnagar, Nimalan Arinaminpathy, et al. Prudent public health intervention strategies to control the coronavirus disease 2019 transmission in India: A mathematical model-based approach. Indian Journal of Medical Research. 2020; 151(2), 190-199.
[48] COV-IND-19 Study Group. Accessed May 2, 2020.
[49] The Center for Disease Dynamics, Economics and Policy. Covid-19 in India. Accessed on May 2, 2020.
[50] Rajesh Singh and R. Adhikari. Age-structured impact of social distancing on the COVID-19 epidemic. arXiv. March 26, 2020.
The views expressed above belong to the author(s). ORF research and analyses now available on Telegram! Click here to access our curated content — blogs, longforms and interviews.

Dr. Erfan Ibrahim has worked in advanced nuclear technology mathematical modeling networking telecommunications smart grid and cybersecurity during his 33-year professional career. He holds a ...
Read More +
Dr. Shahid Jameel established and for 25 years led the Virology Group at the International Centre for Genetic Engineering and Biotechnology New Delhi. He was ...
Read More +