-
CENTRES
Progammes & Centres
Location
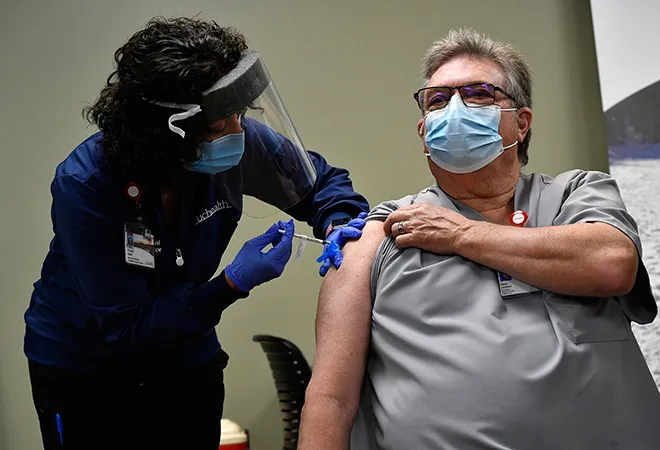
For 300,000 Americans, the country’s first Covid-19 vaccine came too late. For the living, the biggest vaccination campaign in US history kicked off on December 14 as health workers rolled up their sleeves to get the first shots of the Pfizer-BioNTech vaccine which rolled out in a record shattering time frame of 10 months.
From coast to coast, it was a day of collective relief and goosebumps as live television cameras zoomed in on the bare arms of nurses and doctors getting the shots that mark a decisive turn in a catastrophic outbreak.
“I feel hopeful today. Relieved,” critical care nurse Sandra Lindsay said after getting a shot in her left arm at Long Island Jewish Medical Center in New York. “I feel like healing is coming.”
On the same morning, the US crossed 300,000 deaths, based on data from the Johns Hopkins coronavirus tracker. That’s equal to a hundred 9/11 attacks or five times the number of Americans killed in the Vietnam war or the entire population of Pittsburgh. Physicians on the frontlines believe the real number of lives lost is much higher, in part because of deaths that were not accurately recorded during the early months. The seven-day rolling average for daily cases rose to 214,000 on December 13.
The US is hoping to vaccinate 20 million people by December end, comprising healthcare workers and nursing home staff and residents. The average American can expect to get a shot in the “second quarter” of next year, according to Dr. Anthony Fauci, director of the National Institute of Allergy and Infectious Diseases.
Around 3 million shots are out in the first round of shipments. The government is holding back the second dose to ensure that people who got the first shots get the second round. Apart from this total first round of 6.4 million doses, an extra 500,000 doses are being kept for any emergency. States are getting vaccine doses based on population 18 and older.
Assuming the Moderna vaccine also gets regulatory approval, US officials expect they will have enough doses to vaccinate 20 million people by December end - which means they will have 40 million shots available between Pfizer/ BioNTech and Moderna. Moderna’s second shot is timed four weeks after the first.
By February end, there will be enough to vaccinate 100 million Americans, according to data shared by the Trump administration’s vaccine czar Moncef Slaoui.
There are still things we don’t know about the vaccine, which even those who okayed the vaccine don’t know. But the reason they greenlighted it is because what is known outweighs the risk of getting sick with the coronavirus.
For nine hours on December 10, independent experts on the Vaccines and Related Biological Products Advisory Committee of the US Food and Drug Administration (FDA) who represented specialties ranging from pediatrics to virology and ethics debated every piece of information from the Pfizer-BioNTech clinical trial that enrolled 44,000 participants.
For more than two hours in the very end, a single sticking point dominated: vaccine safety in 16 and 17 year olds. One of the panel members urged the others to see that “the data is thinnest in this age group,” and at least three panelists wanted vaccinations to begin only for people older than 18. Those in favor of keeping the 16 year olds in the vaccine’s ambit spoke about how important this group was to the “endgame” and the ability to move the vaccine downwards into younger populations.
The day-long meeting of the FDA vaccine committee meeting shone a light on the questions that will inform the next phase of the ongoing vaccine trials and what to expect. We know now that the most common reactions were pain at the injection site, fever, headache, and fatigue. Most reactions surfaced on the day after vaccination and in Pfizer’s Phase 3 trials, those reactions were more common in younger participants.
Results from the Pfizer-BioNTech trials indicate that even one dose of the vaccine might offer protection, which Pfizer plans to study. Pfizer’s is a two-shot vaccine, the second shot comes three weeks after the first.
Trials for young people between 12-15 years of age are just starting and information on 16 and 17 year olds is very thin because few participated in the trials. Pregnant women were not part of the Pfizer study but 23 women who participated in the study became pregnant during the trial’s duration. There isn’t enough data to say how those women among this cohort who got the vaccine were affected.
In the Pfizer-BioNTech study, 137 (0.63%) of vaccine recipients reported symptoms that suggested an allergic reaction, compared to 111 (0.51% ) in the placebo group.
“It’s never an issue of when do you know everything,” said Dr. Paul Offit, of the Children’s Hospital of Philadelphia. “The question is whether you know enough.” Offit was on the FDA panel debating the Pfizer vaccine.
Finally, the committee voted 17-4 to pave the way for an epic vaccination campaign. On the same day, Donald Trump belted out a counterprogramming stunt at the White House, signing papers prioritizing the “American vaccine” for “American citizens, before sending it to other nations.” On the night of December 11, US regulators approved emergency use of the Pfizer and BioNTech vaccine for people aged 16 years and older.
On December 14, there was only one genre of must-see TV: mass vaccinations. Not Trump.
The views expressed above belong to the author(s). ORF research and analyses now available on Telegram! Click here to access our curated content — blogs, longforms and interviews.

Nikhila Natarajan is Senior Programme Manager for Media and Digital Content with ORF America. Her work focuses on the future of jobs current research in ...
Read More +