-
CENTRES
Progammes & Centres
Location
 Coronavirus patients around the world are lining up to join clinical trials that have opened in hospitals racing to find a breakthrough vaccine against the pandemic. In the US, interest in remdesivir studies has surged. Here, a National Institutes of Health (NIH) study has among the most rigorous protocols in place, comparing remdesivir with placebo infusions. Until the end of the NIH trial, neither patients nor doctors will know who was injected with what. Gilead Sciences, which makes the drug, is going all in with its own trials. Gilead has been studying the potential of remdesivir for many years as part of its antiviral research. This particular medicine is in a more advanced stage of testing in the universe of potential COVID19 therapies.
US President Donald Trump has continued to push his outsize pitch for anti-malarial medicine hydroxychloroquine even though scientists continue to say there is at best only anecdotal evidence of its effectiveness against COVID-19. Hydroxychloroquine is approved for treating malaria, rheumatoid arthritis and lupus, not COVID-19. “What do I know, I’m not a doctor. But I have common sense”, Trump said when asked why he is touting an unapproved drug.
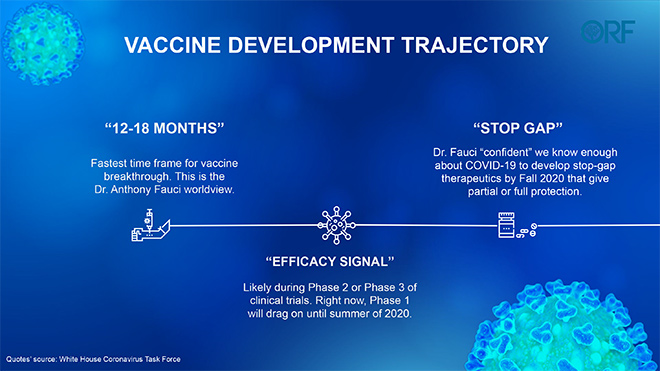
Dr. Anthony Fauci, America’s top infectious diseases doctor-scientist on the White House task force, has cautioned that COVID-19 is showing all the signs that it could hit on a seasonal cycle and that a vaccine is the ultimate gamechanger. According to him, the most aggressive timeline could give the world a vaccine in “12-18 months”. Until the entire country is protected with therapeutics and a vaccine, Fauci says it is unlikely that America can go back to the pre-coronavirus normal.
Based on NIH data, we bring you key highlights about ongoing clinical trials in the race to find a COVID-19 vaccine.
A cure can't come soon enough.
Coronavirus patients around the world are lining up to join clinical trials that have opened in hospitals racing to find a breakthrough vaccine against the pandemic. In the US, interest in remdesivir studies has surged. Here, a National Institutes of Health (NIH) study has among the most rigorous protocols in place, comparing remdesivir with placebo infusions. Until the end of the NIH trial, neither patients nor doctors will know who was injected with what. Gilead Sciences, which makes the drug, is going all in with its own trials. Gilead has been studying the potential of remdesivir for many years as part of its antiviral research. This particular medicine is in a more advanced stage of testing in the universe of potential COVID19 therapies.
US President Donald Trump has continued to push his outsize pitch for anti-malarial medicine hydroxychloroquine even though scientists continue to say there is at best only anecdotal evidence of its effectiveness against COVID-19. Hydroxychloroquine is approved for treating malaria, rheumatoid arthritis and lupus, not COVID-19. “What do I know, I’m not a doctor. But I have common sense”, Trump said when asked why he is touting an unapproved drug.
Dr. Anthony Fauci, America’s top infectious diseases doctor-scientist on the White House task force, has cautioned that COVID-19 is showing all the signs that it could hit on a seasonal cycle and that a vaccine is the ultimate gamechanger. According to him, the most aggressive timeline could give the world a vaccine in “12-18 months”. Until the entire country is protected with therapeutics and a vaccine, Fauci says it is unlikely that America can go back to the pre-coronavirus normal.
Based on NIH data, we bring you key highlights about ongoing clinical trials in the race to find a COVID-19 vaccine.
A cure can't come soon enough.



The views expressed above belong to the author(s). ORF research and analyses now available on Telegram! Click here to access our curated content — blogs, longforms and interviews.

Nikhila Natarajan is Senior Programme Manager for Media and Digital Content with ORF America. Her work focuses on the future of jobs current research in ...
Read More +