-
CENTRES
Progammes & Centres
Location
The global angst against China over allegedly ‘creating’ the coronavirus and then concealing crucial information is still fresh. Widely called out as a ‘charm offensive’ by the Western media, China’s vaccine diplomacy is seen as a means of damage control for its worldwide falling reputation and worsening cross-country ties. Although China’s initial missteps do point to an intentional role in the global pandemic, it is now trying hard to emerge as a dependable ally to its neighbours and countries everywhere who are in dire straits.
Since it started vaccine mass production early this year, China has sent out over 405 million vaccine doses—primarily Sinopharm, CanSino, and Sinovac—to countries everywhere through bilateral agreements via donations and loans. In terms of absolute numbers, the top recipients of Chinese vaccine doses are (including placed orders) – Indonesia (140.5 M), Brazil (100 M), Turkey (100 M), Mexico (67 M), and Pakistan (65.6 M). The Asia-Pacific region continues to be its biggest beneficiary, followed by Latin America.
Image 1: Chinese vaccines distribution across the World
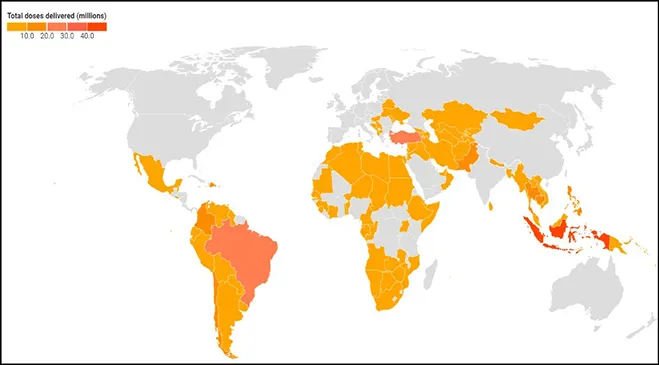 Source: Bridge Beijing
Source: Bridge BeijingChina has grown into a scientific leader in competition with the US and UK with nine vaccines in Phase-3 testing when very few countries have prepared even one. But despite such a display of prowess, its leading vaccines provide relatively low protection compared to their American and European counterparts. Countries such as Seychelles, Bahrain, Mongolia, Chile, etc., who used Chinese vaccines extensively, saw a dangerous second wave. According to Western media, the blame goes to Chinese vaccines Sinopharm and Sinovac.
Below is the list of the top 25 countries currently leading in daily new confirmed cases per million. It is important to note that all countries using Chinese vaccines also use other American-European or Russian vaccines to immunise their population.
Table 1: Top 25 countries with highest daily new confirmed COVID-19 cases per million population as of 3rd July 2021
| Country | Chinese vaccines used | Percentage of the population fully vaccinated (%) | |
| 1 | Seychelles | Sinopharm | 69.29 |
| 2 | Mongolia | Sinopharm | 54.52 |
| 3 | Namibia | Sinopharm | 1.08 |
| 4 | Colombia | Sinovac | 15.84 |
| 5 | Cyprus | NO CHINESE VACCINE USED | 44.52 |
| 6 | Tunisia | Sinovac | 5.15 |
| 7 | Argentina | Sinopharm | 10.74 |
| 8 | Kuwait | NO CHINESE VACCINE USED | 21.62 |
| 9 | Oman | Sinovac | 4.93 |
| 10 | Fiji | NO CHINESE VACCINE USED | 6.11 |
| 11 | UK | NO CHINESE VACCINE USED | 50.38 |
| 12 | South Africa | NO CHINESE VACCINE USED (slated to receive Sinopharm) | 2.11 |
| 13 | Uruguay | Sinovac | 54.65 |
| 14 | Maldives | Sinopharm | 38.16 |
| 15 | Costa Rica | NO CHINESE VACCINE USED | 16.04 |
| 16 | Suriname | NO CHINESE VACCINE USED | 6.70 |
| 17 | Cuba | NO CHINESE VACCINE USED | 14.70 |
| 18 | Panama | NO CHINESE VACCINE USED | 13.64 |
| 19 | Botswana | Sinovac | 4.52 |
| 20 | Brazil | Sinovac | 13.88 |
| 21 | Georgia | Sinovac, Sinopharm | 2.80 |
| 22 | Paraguay | Sinovac, Sinopharm | 1.91 |
| 23 | Kyrgyzstan | Sinopharm | 1.13 |
| 24 | Portugal | NO CHINESE VACCINE USED | 39.18 |
| 25 | Malaysia | Sinovac | 9.86 |
Source: Our World in Data
A broad look at the list clearly shows that the surge is more prevalent in countries using either Sinovac or Sinopharm than non-Chinese manufacturers. Fifteen out of the above 25 countries are users of Chinese vaccines, amounting to 64 percent of all. The percentage continues to remain much more than 60 percent among the top 30 and 45 countries as well if the above list is extended. This raises doubts but does not explicitly imply the causation to be Chinese vaccines only.
On a second note, with a closer look at the trajectories of Chinese vaccine-using countries, post the beginning of immunisation globally (i.e., December 2020), it is found that almost all of them faced a surge. Noticeably, Seychelles, Maldives, and Uruguay saw an abnormal rise compared to others in the group.
Image 2: Trajectories of Chinese-vaccine-Using Countries since 1st December 2020
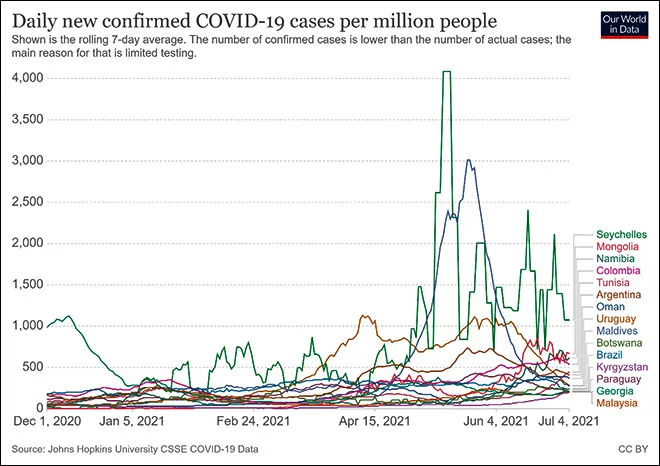 Source: Our World in Data
Source: Our World in DataTable 2: Countries using Chinese Vaccines from the above table – Chinese doses received and Total doses administered (as of 5th July 2021)
| Country | Vaccines used | Total Vaccine doses administered (in millions) | Chinese vaccine doses received (in millions) | Percentage of the population fully vaccinated (%) | |
| 1 | Seychelles | Sinopharm | 0.139 | Un-announced number | 69.29 |
| 2 | Mongolia | Sinopharm | 3.83 | 1.5 | 54.52 |
| 3 | Namibia | Sinopharm | 0.15 | 0.1 | 1.08 |
| 4 | Colombia | Sinovac | 19.44 | 10.47 | 15.84 |
| 5 | Tunisia | Sinovac | 1.84 | 0.7 | 5.15 |
| 6 | Argentina | Sinopharm | 22.44 | 5.909 | 10.74 |
| 7 | Oman | Sinovac | 1.34 | 0.1 | 4.93 |
| 8 | Uruguay | Sinovac | 4.11 | 1.75 | 54.65 |
| 9 | Maldives | Sinopharm | 0.517 | 0.2 | 38.16 |
| 10 | Botswana | Sinovac | 0.26 | 0.4 | 4.52 |
| 11 | Brazil | Sinovac | 106.91 | 57 | 13.88 |
| 12 | Georgia | Sinovac, Sinopharm | 0.37 | 1.2 | 2.80 |
| 13 | Paraguay | Sinovac, Sinopharm | 0.76 | 0.27 | 1.91 |
| 14 | Kyrgyzstan | Sinopharm | 0.173 | 0.15 | 1.13 |
| 15 | Malaysia | Sinovac | 9.32 | 3.5 | 9.86 |
Source: Bridge Beijing and Our World in Data
Interestingly within the above group, countries with a higher percentage of the population vaccinated also faced a higher surge. Several of these countries belong to low and lower-middle-income country groups who were able to kickstart their immunisation program primarily because of the assistance from China. Hence, coupling the lower efficacy of the vaccines with a higher proportion of Chinese vaccines in the whole mix, the defences appear weaker. Additionally, the non-adherence to safe behaviour post inoculation adds to the risk.
Whilst researchers warned against using a vaccine without a large-scale randomised placebo-controlled trial, China had already begun vaccinating soldiers, government employees, aviation staff, and healthcare workers at home.
Amidst global contention, Chinese Government officials have admitted to the comparatively lower efficacy rate. They are exploring the possibility of mixing two vaccines or adding booster shots to reach promised protection. But Chinese officials have also denied the efficacy argument to be the prime reason for breakout infections, and instead point to the non-compliance to COVID-appropriate behaviour and the emergence of new, more transmissible variants as glaring reasons for disquietude.
While the world was still figuring out the coronavirus, China led an ambitious scientific project by having the most vaccines under development (11 out of 42 vaccines in September 2020). It was among the first few countries which came up with a vaccine the fastest. Its immunisation initiative started as early as 25 June 2020, with CanSino's single-dose vaccine's emergency approval. By July 2020, the country already had two vaccines approved for emergency usage. Whilst researchers warned against using a vaccine without a large-scale randomised placebo-controlled trial, China had already begun vaccinating soldiers, government employees, aviation staff, and healthcare workers at home.
As a show of great confidence, by December 2020, the UAE and Bahrain granted Sinopharm full clearance after indicating 86 percent efficacy in the trial results, but without ever disclosing the data. Six months later, now, after vaccinating 157 and 126 per 100 people respectively (among the highest in the world), they are offering booster shots. The boosters are only for Sinopharm-vaccinated citizens merely six months after completing its two-dose schedule and after three months for the high-risk category population. Concrete data on the effectiveness of the subsequent booster shots is still unavailable.
More so, the clinical trial designed to study its efficacy consisted of an essentially healthy male-dominated group with a median age of 36 years. Hence, the efficacy of 79 percent is not a reliable indicator for women, asymptomatic cases, or the elderly population of 60+ years. The WHO’s assessment of the trial evidence also admits to a very low level of confidence in its capacity to prevent severe adverse events in people with long-term diseases. A corroborating real-world study was reported by the Wall Street Journal, which stated that 29 percent of its recipients from the 60+ years age group in Serbia had zero antibodies against the virus.
Drug’s protection towards the elderly population of 60+ years and the ones with long-term comorbidities is uncertain.
Sinovac's CoronaVac also faced criticism when it yielded strikingly low yet fluctuating efficacy rates in different countries—51 percent in Brazil, 65.3 percent in Indonesia, 67 percent in Chile, and 83.5 percent in Turkey. Even the highest reported efficacy from the trials, i.e., in Turkey, is not actively generalisable. The study group consisted of a relatively young population with a median age of 45 years and a low prevalence of chronic illnesses, unlike Pfizer and Moderna. The studies’ protection time for both Sinopharm and Sinovac after vaccinating with both doses was a mere 112 and 43 days, respectively. Hence, eventually implying that the drug’s protection towards the elderly population of 60+ years and the ones with long-term comorbidities is uncertain. Adding to the bargain, earlier this year, the CanSino Biologics also claimed that their vaccine's efficacy would likely fall to 50 percent just after five to six months of vaccination and would require booster shots to maintain the promised effectiveness.
While the second wave was a common feature in nearly all countries from February to June 2021, including India, but such a rise in cases was not an expected outcome from countries closer to herd immunity. The initial assessment indicates blame on the poor shielding against the coronavirus offered by Chinese vaccines. It seems like the Chinese government was too quick to approve the vaccines without all available data from trials to mend the Communist Party’s initial lapses.
China has been contesting appeal for its home-grown vaccines globally through vaccine diplomacy. But when Xi Jinping called its vaccines 'a global public good', its approach and pricing towards the countries have been biased. Reports of its diplomacy being preferential towards some have caused much stir. Presumably, Sri Lanka is paying US$ 15 per dose for Sinopharm, while Nepal and Bangladesh are paying only US$ 10 per dose.
Experts have noted that despite lower efficacies, the Chinese vaccines have successfully lowered COVID-19 hospitalisations and deaths, hence serving the true purpose of a vaccine.
In addition, it recently instituted stringent visa moderations favouring only those inoculated with its vaccines. Hence, citizens of the neighbouring regions of Hong Kong, Singapore, Vietnam, Indonesia, etc., despite having choices, will be compelled to choose a Chinese vaccine to ensure easy mobility in and around Mainland China.
What provides ascendency to Sinopharm and Sinovac in the global south and what has also been a tactful advantage to China is that they use a more established and time-tested platform of inactivated viruses to combat the infection. This makes them preferable among developing economies as they are more suited for basic health infrastructures that do not require end-to-end refrigeration. Therefore, despite questions on and acrimony around the Chinese brand, the demand for these vaccines is still growing, counting for more than 500 million pre-ordered doses. The World Health Organisation granted Emergency Listing to these two vaccines in May and June 2021, thereby, giving a mighty push to their demand. Considering that the country alone has administered 1.35 billion doses at home, its massive vaccine manufacturing capacity can provide a much-needed boost for the cause of vaccine equity in the wake of global shortage and easing the global overdependency on the Serum Institute for vaccines.
Amidst widespread vaccine hesitancy, the underperformance of these vaccines can prove antagonistic to the immunisation program. With severe global distrust already looming over China's intentions, scientific peer support is essential to its credibility as a major vaccine manufacturer. Without reliable data on their real-world effectiveness, it would still be too early to make verdicts. Experts have noted that despite lower efficacies, the Chinese vaccines have successfully lowered COVID-19 hospitalisations and deaths, hence serving the true purpose of a vaccine. Seychelles has defended the vaccines reporting that about 80 percent of the deaths and severe hospitalisations happened with the population either partly or not vaccinated.
Hence, it is prime to yield some benefit of the doubt to Chinese manufacturers in the absence of complete, real-world information on their effectiveness and its emerging role as an equity champion. After all, it is a race that can only be won together.
The views expressed above belong to the author(s). ORF research and analyses now available on Telegram! Click here to access our curated content — blogs, longforms and interviews.

Mona is a Junior Fellow with the Health Initiative at Observer Research Foundation’s Delhi office. Her research expertise and interests lie broadly at the intersection ...
Read More +